Jeil Pharm, Shin Poong Pharm actively invest in R&D for 2 trillion won market
KIST, Celltrion dives into market with industry-academic collaboration research
According to the World Health Organization, stroke is the second leading cause of death, with the global market for ischemic stroke medicines expected to reach about 2.3 trillion won this year.
Against this backdrop, the domestic pharmaceutical industry is gaining momentum in researching and developing local stroke treatments.
Jeil Pharm and Shin Poong Pharm have begun developing new stroke treatments. At the same time, others such as KIST and Celltrion joined hands in industry-academia-based cooperation to develop them.
Korean pharma makes strikes in developing stroke treatments
Stroke therapy is historically challenging to make, with most failing in clinical trials. It’s also challenging to determine the safety, efficacy, and toxicity of new drug candidates since stroke itself exacerbates the patient’s condition.
For these reasons, multinational pharmaceuticals have reported having faced difficulties in identifying and developing new drug candidates.
The only drug currently used to treat stroke is Boehringer Ingelheim’s tissue-type plasminogen activator (tPA) activase (ingredient alteplase), an intravenous hemolytic drug. However, tPA activase is limited in that the drug’s efficacy and side effects may occur if not administered within three to four hours of stroke
Korean pharma, noting the lack of treatments in the market, have become active in researching and developing stroke drugs.
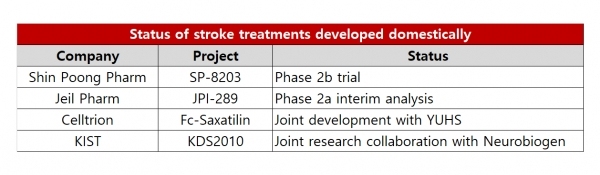
Jeil Pharm is developing stroke treatment JPI-289. JPI-289 inhibits the DNA damage caused by cerebral ischemia and PARP enzymes involved in killing nerve cells.
Jeil Pharm is currently performing an interim analysis after completing two phase 2a cohort studies.
Cohort 1 included 15 patients, and cohort 2 included 15 patients, bringing the total of patients in the phase 2a study to 30. Jeil said it confirmed the effect of reducing cerebral infarct volume at 90 days in the JPI-289 arm compared to the placebo arm.
JPI-289 also showed improvement of one point on the six-point disability scale that is the modified Ranking Scale (mRS).
Based on the results, Jeil said the Ministry of Food and Drug Safety approved the cohort 3 protocol, which is the final step of the phase 2a study.
Shin Poong is also conducting a Phase 2b trial for SP-8203, a new stroke treatment.
SP-8203 is a mechanism that inhibits brain hemorrhage like tPA, by increasing TIMP and decreasing matrix metalloproteinase (MMP) enzyme activity.
SP-8203 can be used in combination with a monotherapy. In particular, SP-8203, when used in conjunction with tPA, suppressed the side effects caused by tPA, the company said.
Trial results also showed that intracranial hemorrhage, adverse drug reaction rate, and mortality were not significantly between SP-8203 and placebo.
Shin Poong plans to proceed with the phase 1 bridging trial in Europe and the U.S. after deriving research results while launching phase 3 trials in Korea in the second half of this year.
The company also said it would complete phase 3 clinical trials by the end of 2022 and complete the new drug application (NDA) in 2023.
Celltrion-KIST stroke collaboration fires up industry-academy cooperation
Industry-academic cooperation for the development of stroke medicines is also gaining traction in the country.
Celltrion signed a contract with Yonsei University Health System last year for joint development and licensing of Fc-Saxatilin, a candidate for ischemic stroke.
The candidate drug is a new substance developed by the Yonsei Integrative Research Institute for Cerebral & Cardiovascular Disease under the support of the Ministry of Health and Welfare.
Industry insiders expect the drug to show superior thrombolytic effect and fewer side effects in patients with ischemic stroke.
Celltrion and Yonsei University Health System will jointly develop the candidate drug, maximizing the collaboration potential. Celltrion plans to carry out clinical trials, approvals, and commercialization with a global license.
Recently the Korea Institute of Science and Technology (KIST) completed a technology transfer and joint research and development (R&D) agreement with Neurobiogen to develop drugs that block the excessive production of a neurotransmitter called GABA. GABA is a signaling substance that inhibits neuronal activity in the central nervous system.
The KIST team led by Dr. Park Ki-duk previously developed KDS2010, a substance that prevents excessive production of GABA.
KIST identified the possibility that KDS2010 could reverse the motor function in stroke animal models. The institution then joined hands with the company to develop the candidate as a therapeutic drug for spinal cord injury and stroke.
The two parties plan on conducting a phase 1 study after completing the preclinical trial this year.
“tPA activase is the only treatment in the stroke market, and companies are keen to enter the market as a latecomer,” an industry official said. “Multinational pharmaceuticals gained a head start in developing stroke treatments, but Korean companies are expected to close the gap considerably as they are making significant R&D achievements.”